
Carbon Monoxide 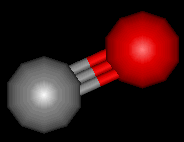 
Dr Mike Thompson
Winchester College, UK 
Also available: Chime Enhanced, VRML and JMol versions.

Carbon monoxide is a toxic, colourless and odourless gas. It has been invaluable in helping chemists to extract metals from their ores. However, it must be said that its physical properties make it potentially very dangerous.
Making Carbon Monoxide Carbon and oxygen can combine to form two gases. When combustion of carbon is complete, i.e. in the presence of plenty of air, the product is mainly carbon dioxide (CO2). Sources of carbon include; coal, coke, charcoal. When combustion of carbon is incomplete, i.e. there is a limited supply of air, only half as much oxygen adds to the carbon, and instead you form carbon monoxide (CO).
 Carbon monoxide is also formed as a pollutant when hydrocarbon fuels (natural gas, petrol, diesel) are burned. The relative amount of CO produced depends on the efficiency of combustion. Older vehicles are checked annually for CO emissions during their MOT test. Interestingly, only one of the two oxides of carbon, doesn't support combustion and it is for this reason that carbon dioxide is used in fire extinguishers. Carbon monoxide does support combustion and burns with a pale blue flame. The blue flame used to be seen over the fires made from coke (essentially a very pure form of carbon) by night watchmen on industrial sites. Carbon monoxide is also formed as a pollutant when hydrocarbon fuels (natural gas, petrol, diesel) are burned. The relative amount of CO produced depends on the efficiency of combustion. Older vehicles are checked annually for CO emissions during their MOT test. Interestingly, only one of the two oxides of carbon, doesn't support combustion and it is for this reason that carbon dioxide is used in fire extinguishers. Carbon monoxide does support combustion and burns with a pale blue flame. The blue flame used to be seen over the fires made from coke (essentially a very pure form of carbon) by night watchmen on industrial sites.
2 CO (g) + O2 (g) 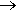 2 CO2 (g) 2 CO2 (g)
Laboratory Preparation of Carbon monoxide From Carbon: When making carbon monoxide a source of carbon dioxide gas is needed. This could be from a CO2 cylinder or even dry ice (solid CO2). If neither is available carbon dioxide could be generated by the neutralisation reactions between an acid and a carbonate or an acid and a hydrogen carbonate.
2HCl (aq) + CaCO3 (s) 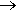 CaCl2 (aq) + H2O (l) + CO2 (g) CaCl2 (aq) + H2O (l) + CO2 (g)
HCl (aq) + NaHCO3 (s) 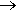 NaCl (aq) + H2O (l) + CO2 (g) NaCl (aq) + H2O (l) + CO2 (g)
When carbon dioxide gas is passed over heated charcoal it forms carbon monoxide.
CO2 (g) + C (s) 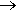 2CO (g) 2CO (g)
There will also be unreacted carbon dioxide, which needs to be removed. Carbon dioxide is removed by reacting it with an aqueous solution of sodium hydroxide.
2 NaOH (aq) + CO2 (g) 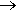 Na2CO3 (aq) + H2O (l) Na2CO3 (aq) + H2O (l)
From Methanoic Acid: Another convenient way to prepare carbon monoxide is the dehydration of methanoic acid using conc. H2SO4.
HCOOH (aq) 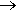 CO (g) + H2O (l) CO (g) + H2O (l)
Dehydration of a methanoic salt such as sodium methanoate also works well. In this case you dribble concentrated sulphuric acid directly onto the solid. The carbon monoxide, which evolves, can be collected under water. Methanoic acid is found in nettles and ants.
How poisonous?  Carbon monoxide is a very poisonous gas. It is poisonous at levels of only 0.1% (1000 ppm). Its toxicity arises from its ability to bind to transition metals such as iron found at the centre of a haem molecule. Carbon monoxide is attracted to haemoglobin over 200 times more strongly than oxygen. Therefore, in the blood, the presence of carbon monoxide prevents some of the haemoglobin found in red blood cells from carrying sufficient oxygen. Carbon monoxide is a very poisonous gas. It is poisonous at levels of only 0.1% (1000 ppm). Its toxicity arises from its ability to bind to transition metals such as iron found at the centre of a haem molecule. Carbon monoxide is attracted to haemoglobin over 200 times more strongly than oxygen. Therefore, in the blood, the presence of carbon monoxide prevents some of the haemoglobin found in red blood cells from carrying sufficient oxygen.
This fact is certainly worth considering if you should be tempted to smoke a cigarette. Smokers have been found to have quite high levels of carbon monoxide in their blood, long after they have finished lighting up their chosen cigarette.
 Symptoms of carbon monoxide poisoning are dizziness and headaches. These sufferings can be confused with other illnesses such as influenza. Carbon monoxide poisoning can be recognised, as victims will often have unnaturally bright red lips. Symptoms of carbon monoxide poisoning are dizziness and headaches. These sufferings can be confused with other illnesses such as influenza. Carbon monoxide poisoning can be recognised, as victims will often have unnaturally bright red lips.
Prolonged exposure to carbon monoxide can eventually lead to death. Carbon monoxide has been used as the poison in suicides. Even more disturbing was the use of carbon monoxide by Nazis in WWII to kill its victims of the death camps. More recently, there have been cases of rogue landlords not properly maintaining gas appliances, which led to the death of tenants often students. The law in the UK now requires the annual checking of boilers, gas cookers, and gas fires by registered engineers. The following equation shows what happens when there is incomplete combustion of natural gas, which is primarily methane.
2 CH4 (g) + 3 O2 (g) 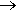 2 CO (g) + 4 H2O (g) 2 CO (g) + 4 H2O (g)
Coal Gas  Before vast quantities of natural gas were discovered beneath the seas and oceans, we used to burn coal gas. Coal gas was produced when coal was heated in the absence of air. Its main components are hydrogen methane, and carbon monoxide. Before vast quantities of natural gas were discovered beneath the seas and oceans, we used to burn coal gas. Coal gas was produced when coal was heated in the absence of air. Its main components are hydrogen methane, and carbon monoxide.
Carbon monoxide sometimes occurs in coal mines. At one time canaries were taken down in mines to detect poisonous gases. Canaries would be killed at doses not quite lethal to miners. Today gases are detected more humanely with instruments.
Reducing agent 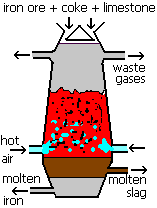 The first time most students meet carbon monoxide in a chemistry lesson is in its use within the Blast Furnace. The Blast furnace is how iron is extracted from its ore, haematite (iron(III)oxide Fe2O3). The first time most students meet carbon monoxide in a chemistry lesson is in its use within the Blast Furnace. The Blast furnace is how iron is extracted from its ore, haematite (iron(III)oxide Fe2O3).
Fe2O3 (s) + 3 CO (g) 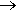 2 Fe (l) + 3 CO2 (g) 2 Fe (l) + 3 CO2 (g)
Carbon monoxide is a strong reducing agent and reduces metal oxides for metals less reactive than carbon. The following table is useful for the different definitions of reduction. As oxidation is the opposite of reduction you only need to learn half the facts!
Reduction Oxidation | Loss of oxygen | Gain of oxygen | | Gain of hydrogen | Loss of hydrogen | | Gain of electrons | Loss of electrons | | Decrease in O.N. | Increase in O.N. | Reactions of Carbon monoxide Several gases (H2, CH4 and CO) have historically been used as reducing agents. One definition of a reducing agent I particularly like is to think of it as being an oxygen grabber. It is important to remember that the reducing agent itself gets oxidised. A quick and simple laboratory reduction can be achieved by heating a mixture of black copper(II)oxide with carbon powder in a test tube. After several minutes of heating the reddish coloured copper can be seen on the side of the test tube. Essentially carbon acts as a reducing agent as well as the carbon monoxide that is inevitably formed by its heating in air. The following reactions are all occurring in this simple experiment.
CuO (s) + CO (g) 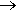 Cu (s) + CO2 (g) Cu (s) + CO2 (g)
CuO (s) + C (s) 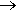 Cu (s) + CO (g) Cu (s) + CO (g)
C (s) + O2 (g) 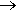 2 CO (g) 2 CO (g)
C (s) + O2 (g) 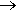 CO2 (g) CO2 (g)
CO2 (g) + C (s) 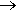 2 CO (g) 2 CO (g)
Affinity for Transition Metals Carbon monoxide does not show acidic or basic properties. Its feeble Lewis acidity is seen through its formation of H3BCO with borane (BH3). Carbon monoxide has a remarkable affinity for transition metals (located between Groups 2 and 3 of the Periodic Table). The first examples of metal carbonyls was back in 1888, when tetracarbonyl nickel (0) Ni(CO)4 and pentacarbonyl iron (0) Fe(CO)5 were prepared and characterised. The former complex forms part of the Mond Process for the purification of Nickel. Ni(CO)4 is distilled to give pure nickel.
Ni (s) + 4 CO (g) 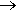 Ni(CO)4 Ni(CO)4 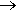 Ni (s) + 4 CO (g) Ni (s) + 4 CO (g)
Carbon monoxide is so reactive with nickel that within a couple of minutes it will have etched the surface. Ni(CO)4 is highly toxic with a musty smell. As well as being flammable this tetrahedral complex decomposes easily into its constituents. Carbon monoxide is acting as a ligand towards the transition metal through the lone pair on the carbon atom. Diatomic carbon monoxide has a triple bond between its atoms. One of the bonds forming the triple bond is a dative covalent bond.
 Please try this at home Detecting carbon monoxide is a simple task these days. Most hardware stores sell special spots impregnated with palladium compounds, which darken on exposure to carbon monoxide. Please try this at home Detecting carbon monoxide is a simple task these days. Most hardware stores sell special spots impregnated with palladium compounds, which darken on exposure to carbon monoxide.

 Back to Molecule of the Month page. Back to Molecule of the Month page.
Heat of combustion
From Wikipedia, the free encyclopedia
Jump to: navigation, search
The heat of combustion ( ) is the energy released as heat when a compound undergoes complete combustion with oxygen under standard conditions. The chemical reaction is typically a hydrocarbon reacting with oxygen to form carbon dioxide, water and heat. It may be expressed with the quantities: ) is the energy released as heat when a compound undergoes complete combustion with oxygen under standard conditions. The chemical reaction is typically a hydrocarbon reacting with oxygen to form carbon dioxide, water and heat. It may be expressed with the quantities:
- energy/ mole of fuel ( kJ/mol)
- energy/mass of fuel
- energy/volume of fuel
The heat of combustion is conventionally measured with a bomb calorimeter. It may also be calculated as the difference between the heat of formation  of the products and reactants. of the products and reactants.
Contents[ hide]
Heating value[ edit] The heating value (or energy value or calorific value) of a substance, usually a fuel or food (see food energy), is the amount of heat released during the combustion of a specified amount of it. The energy value is a characteristic for each substance. It is measured in units of energy per unit of the substance, usually mass, such as: kJ/kg, kJ/ mol, kcal/kg, Btu/lb. Heating value is commonly determined by use of a bomb calorimeter.
Heating value unit conversions (for more visit Wolfram Alpha):
- kcal/kg = MJ/kg * 238.846
- Btu/lb = MJ/kg * 429.923
- Btu/lb = kcals * 1.8
The heat of combustion for fuels is expressed as the HHV, LHV, or GHV.
Higher heating value[ edit] The quantity known as higher heating value (HHV) (or gross energy or upper heating value or gross calorific value (GCV) or higher calorific value (HCV)) is determined by bringing all the products of combustion back to the original pre-combustion temperature, and in particular condensing any vapor produced. Such measurements often use a standard temperature of 25°C. This is the same as the thermodynamic heat of combustion since the enthalpy change for the reaction assumes a common temperature of the compounds before and after combustion, in which case the water produced by combustion is liquid.
The higher heating value takes into account the latent heat of vaporization of water in the combustion products, and is useful in calculating heating values for fuels where condensation of the reaction products is practical (e.g., in a gas-fired boiler used for space heat). In other words, HHV assumes all the water component is in liquid state at the end of combustion (in product of combustion) and that heat below 150°C can be put to use.
Lower heating value[ edit] The quantity known as lower heating value (LHV) (net calorific value (NCV) or lower calorific value (LCV)) is determined by subtracting the heat of vaporization of the water vapor from the higher heating value. This treats any H2O formed as a vapor. The energy required to vaporize the water therefore is not released as heat.
LHV calculations assume that the water component of a combustion process is in vapor state at the end of combustion, as opposed to the higher heating value (HHV) (a.k.a. gross calorific value or gross CV) which assumes that all of the water in a combustion process is in a liquid state after a combustion process.
The LHV assumes that the latent heat of vaporization of water in the fuel and the reaction products is not recovered. It is useful in comparing fuels where condensation of the combustion products is impractical, or heat at a temperature below 150°C cannot be put to use.
The above is but one definition of lower heating value adopted by the American Petroleum Institute (API) and uses a reference temperature of 60°F (15.56°C).
Another definition, used by Gas Processors Suppliers Association (GPSA) and originally used by API (data collected for API research project 44), is the enthalpy of all combustion products minus the enthalpy of the fuel at the reference temperature (API research project 44 used 25°C. GPSA currently uses 60°F), minus the enthalpy of the stoichiometric oxygen (O2) at the reference temperature, minus the heat of vaporization of the vapor content of the combustion products.
The distinction between the two is that this second definition assumes that the combustion products are all returned to the reference temperature and the heat content from the condensing vapor is considered not to be useful. This is more easily calculated from the higher heating value than when using the preceding definition and will in fact give a slightly different answer.
Gross heating value[ edit] - Gross heating value (see AR) accounts for water in the exhaust leaving as vapor, and includes liquid water in the fuel prior to combustion. This value is important for fuels like wood or coal, which will usually contain some amount of water prior to burning.
- Note that GPSA 12th Edition states that the Gross Heating Value of a gas is equivalent to Higher Heating Value. This suggests that there may be different standards in play. The use of the term Gross normally describes a larger value than the Net, which usually describes a smaller value. GPSA is consistent with this, and equates the Gross Heating Value to the higher heating value (for a gas - so probably with no liquid water present), and the Net Heating Value to the lower heating value.
Measuring heating values[ edit] The higher heating value is experimentally determined in a bomb calorimeter. The combustion of a stoichiometric mixture of fuel and oxidizer (e.g., two moles of hydrogen and one mole of oxygen) in a steel container at 25° is initiated by an ignition device and the reactions allowed to complete. When hydrogen and oxygen react during combustion, water vapor is produced. The vessel and its contents are then cooled to the original 25°C and the higher heating value is determined as the heat released between identical initial and final temperatures.
When the lower heating value (LHV) is determined, cooling is stopped at 150°C and the reaction heat is only partially recovered. The limit of 150°C is an arbitrary choice.
Note: Higher heating value (HHV) is calculated with the product of water being in liquid form while lower heating value (LHV) is calculated with the product of water being in vapor form.
Relation between heating values[ edit] The difference between the two heating values depends on the chemical composition of the fuel. In the case of pure carbon or carbon monoxide, the two heating values are almost identical, the difference being the sensible heat content of carbon dioxide between 150°C and 25°C ( sensible heat exchange causes a change of temperature. In contrast, latent heat is added or subtracted for phase transitions at constant temperature. Examples: heat of vaporization or heat of fusion). For hydrogen the difference is much more significant as it includes the sensible heat of water vapor between 150°C and 100°C, the latent heat of condensation at 100°C, and the sensible heat of the condensed water between 100°C and 25°C. All in all, the higher heating value of hydrogen is 18.2% above its lower heating value (142 MJ/kg vs. 120 MJ/kg). For hydrocarbons the difference depends on the hydrogen content of the fuel. For gasoline and diesel the higher heating value exceeds the lower heating value by about 10% and 7% respectively, and for natural gas about 11%.
A common method of relating HHV to LHV is:
HHV = LHV + hv x (nH2O,out/nfuel,in) where hv is the heat of vaporization of water, nH2O,out is the moles of water vaporized and nfuel,in is the number of moles of fuel combusted. [1] Most applications that burn fuel produce water vapor, which is unused and thus wastes its heat content. In such applications, the lower heating value is generally used to give a 'benchmark' for the process; however, for true energy calculations the higher heating value is correct. This is particularly relevant for natural gas, whose high hydrogen content produces much water. The gross energy value is relevant for gas burned in condensing boilers and power plants with flue-gas condensation that condense the water vapor produced by combustion, recovering heat which would otherwise be wasted.
Usage of terms[ edit] For historical reasons, the efficiency of power plants and combined heat and power plants in Europe may have once been calculated based on the LHV. However, this does not seem to be the case nowadays and most countries are tending to correctly use HHV for true efficiency figures. This is becoming noticeable in modern UK energy publications with the increase of energy awareness and based on the simple fact that it is correct. While in the US, values may have been reported to be generally based on the HHV, although any initial investigation may reveal that the US is still tending to use LHV in some circumstances, whether technically correct or not. This has the peculiar result that contemporary combined heat and power plants, where flue-gas condensation is implemented, may report efficiencies exceeding 100%. Using LHV in other energy calculations brings similar errors, especially when pulled (incorrectly) into electrolysis calculations etc.
Many engine manufacturers rate their engine fuel consumption by the lower heating values. American consumers should be aware that the corresponding fuel-consumption figure based on the higher heating value will be somewhat higher.
The difference between HHV and LHV definitions causes endless confusion when quoters do not bother to state the convention being used. [2] since there is typically a 10% difference between the two methods for a power plant burning natural gas. For simply benchmarking part of a reaction the LHV may be appropriate, but HHV should be used for overall energy efficiency calculations, if only to avoid confusion, and in any case the value or convention should be clearly stated.
Accounting for moisture[ edit] Both HHV and LHV can be expressed in terms of AR (all moisture counted), MF and MAF (only water from combustion of hydrogen). AR, MF, and MAF are commonly used for indicating the heating values of coal:
- AR (As Received) indicates that the fuel heating value has been measured with all moisture and ash forming minerals present.
- MF (Moisture Free) or Dry indicates that the fuel heating value has been measured after the fuel has been dried of all inherent moisture but still retaining its ash forming minerals.
- MAF (Moisture and Ash Free) or DAF (Dry and Ash Free) indicates that the fuel heating value has been measured in the absence of inherent moisture and ash forming minerals.
Heat of combustion tables[ edit] Higher (HHV) and Lower (LHV) Heating values
of some common fuels [3] Fuel HHV MJ/kg HHV BTU/lb HHV kJ/ mol LHV MJ/kg | Hydrogen | 141.80 | 61,000 | 286 | 119.96 | | Methane | 55.50 | 23,900 | 889 | 50.00 | | Ethane | 51.90 | 22,400 | 1,560 | 47.80 | | Propane | 50.35 | 21,700 | 2,220 | 46.35 | | Butane | 49.50 | 20,900 | 2,877 | 45.75 | | Pentane | | | | 45.35 | | Gasoline | 47.30 | 20,400 | | 44.4 | | Paraffin wax | 46.00 | 19,900 | | 41.50 | | Kerosene | 46.20 | 19,862 | | 43.00 | | Diesel | 44.80 | 19,300 | | 43.4 | | Coal ( Anthracite) | 32.50 | 14,000 | | | | Coal ( Lignite) | 15.00 | 8,000 | | | | Wood (MAF) | 21.7 | 8,700 | | | | Peat (damp) | 6.00 | 2,500 | | | | Peat (dry) | 15.00 | 6,500 | | | Higher heating value
of some less common fuels [3] Fuel HHV MJ/kg BTU/lb kJ/ mol | Methanol | 22.7 | 9,800 | 726.0 | | Ethanol | 29.7 | 12,800 | 1,300.0 | | 1-Propanol | 33.6 | 14,500 | 2,020.0 | | Acetylene | 49.9 | 21,500 | 1,300.0 | | Benzene | 41.8 | 18,000 | 3,270.0 | | Ammonia | 22.5 | 9,690 | 382.0 | | Hydrazine | 19.4 | 8,370 | 622.0 | | Hexamine | 30.0 | 12,900 | 4,200.0 | | Carbon | 32.8 | 14,100 | 393.5 |
Heat of Combustion for some common fuels (higher value)[ citation needed] Fuel kJ/g kcal/g BTU/lb | Hydrogen | 141.9 | 33.9 | 61,000 | | Gasoline | 47.0 | 11.3 | 20,000 | | Diesel | 45.0 | 10.7 | 19,300 | | Ethanol | 29.7 | 7.1 | 12,000 | | Propane | 49.9 | 11.9 | 21,000 | | Butane | 49.2 | 11.8 | 21,200 | | Wood | 15.0 | 3.6 | 6,000 | | Coal ( Lignite) | 15.0 | 4.4 | 8,000 | | Coal ( Anthracite)[ citation needed] | 27.0 | 7.8 | 14,000 | | Natural Gas | 54.0 | 13.0 | 23,000 |
Lower heating value for some organic compounds (at 15.4°C)[ citation needed][ edit] Fuel MJ/kg MJ/ L BTU/lb kJ/ mol Alkanes | Methane | 50.009 | — | 21,504 | 802.34 | | Ethane | 47.794 | — | 20,551 | 1,437.2 | | Propane | 46.357 | — | 19,934 | 2,044.2 | | Butane | 45.752 | — | 19,673 | 2,659.3 | | Pentane | 45.357 | 28.39 | 21,706 | 3,272.6 | | Hexane | 44.752 | 29.30 | 19,504 | 3,856.7 | | Heptane | 44.566 | 30.48 | 19,163 | 4,465.8 | | Octane | 44.427 | — | 19,104 | 5,074.9 | | Nonane | 44.311 | 31.82 | 19,054 | 5,683.3 | | Decane | 44.240 | 33.29 | 19,023 | 6,294.5 | | Undecane | 44.194 | 32.70 | 19,003 | 6,908.0 | | Dodecane | 44.147 | 33.11 | 18,983 | 7,519.6 | Isoparaffins | Isobutane | 45.613 | — | 19,614 | 2,651.0 | | Isopentane | 45.241 | 27.87 | 19,454 | 3,264.1 | | 2-Methylpentane | 44.682 | 29.18 | 19,213 | 6,850.7 | | 2,3-Dimethylbutane | 44.659 | 29.56 | 19,203 | 3,848.7 | | 2,3-Dimethylpentane | 44.496 | 30.92 | 19,133 | 4,458.5 | | 2,2,4-Trimethylpentane | 44.310 | 30.49 | 19,053 | 5,061.5 | Naphthenes | Cyclopentane | 44.636 | 33.52 | 19,193 | 3,129.0 | | Methylcyclopentane | 44.636? | 33.43? | 19,193? | 3,756.6? | | Cyclohexane | 43.450 | 33.85 | 18,684 | 3,656.8 | | Methylcyclohexane | 43.380 | 33.40 | 18,653 | 4,259.5 | Monoolefins | Ethylene | 47.195 | — | — | — | | Propylene | 45.799 | — | — | — | | 1-Butene | 45.334 | — | — | — | | cis-2-Butene | 45.194 | — | — | — | | trans-2-Butene | 45.124 | — | — | — | | Isobutene | 45.055 | — | — | — | | 1-Pentene | 45.031 | — | — | — | | 2-Methyl-1-pentene | 44.799 | — | — | — | | 1-Hexene | 44.426 | — | — | — | Diolefins | 1,3-Butadiene | 44.613 | — | — | — | | Isoprene | 44.078 | - | — | — | Nitrous derivated | Nitromethane | 10.513 | — | — | — | | Nitropropane | 20.693 | — | — | — | Acetylenes | Acetylene | 48.241 | — | — | — | | Methylacetylene | 46.194 | — | — | — | | 1-Butyne | 45.590 | — | — | — | | 1-Pentyne | 45.217 | — | — | — | Aromatics | Benzene | 40.170 | — | — | — | | Toluene | 40.589 | — | — | — | | o-Xylene | 40.961 | — | — | — | | m-Xylene | 40.961 | — | — | — | | p-Xylene | 40.798 | — | — | — | | Ethylbenzene | 40.938 | — | — | — | | 1,2,4-Trimethylbenzene | 40.984 | — | — | — | | Propylbenzene[ disambiguation needed] | 41.193 | — | — | — | | Cumene | 41.217 | — | — | — | Alcohols | Methanol | 19.930 | 15.78 | 8,570 | 638.55 | | Ethanol | 28.865 | 22.77 | 12,412 | 1,329.8 | | 1-Propanol | 30.680 | 24.65 | 13,192 | 1,843.9 | | Isopropanol | 30.447 | 23.93 | 13,092 | 1,829.9 | | n-Butanol | 33.075 | 26.79 | 14,222 | 2,501.6 | | Isobutanol | 32.959 | 26.43 | 14,172 | 2,442.9 | | Tert-butanol | 32.587 | 25.45 | 14,012 | 2,415.3 | | n-Pentanol | 34.727 | 28.28 | 14,933 | 3,061.2 | | Isoamyl alcohol | 31.416? | 35.64? | 13,509? | 2,769.3? | Ethers | Methoxymethane | 28.703 | — | 12,342 | 1,322.3 | | Ethoxyethane | 33.867 | 24.16 | 14,563 | 2,510.2 | | Propoxypropane | 36.355 | 26.76 | 15,633 | 3,568.0 | | Butoxybutane | 37.798 | 28.88 | 16,253 | 4,922.4 | Aldehydes and ketones | Methanal | 17.259 | — | — | — | | Ethanal | 24.156 | — | — | — | | Propionaldehyde | 28.889 | — | — | — | | Butyraldehyde | 31.610 | — | — | — | | Acetone | 28.548 | 22.62 | — | — | Other species | Carbon (graphite) | 32.808 | — | — | — | | Hydrogen | 120.971 | — | 52,017 | 244 | | Carbon monoxide | 10.112 | — | 4,348 | 283.24 | | Ammonia | 18.646 | — | 8,018 | 317.56 | | Sulfur (solid) | 9.163 | — | 3,940 | 293.82 | Note that there is no difference between the lower and higher heating values for the combustion of carbon, carbon monoxide and sulfur since no water is formed in combusting those substances. BTU/lb values are calculated from MJ/kg (1 MJ/kg = 430 BTU/lb).
Higher heating values of natural gases from various sources[ edit] The International Energy Agency reports the following typical higher heating values: [4]
The lower heating value of natural gas is normally about 90 percent of its higher heating val.
See also[ edit] References[ edit]
Jump up ^ Air Quality Engineering, CE 218A, W. Nazaroff and R. Harley, University of California Berkeley, 2007 Jump up ^ http://www.claverton-energy.com/the-difference-between-lcv-and-hcv-or-lower-and-higher-heating-value-or-net-and-gross-is-clearly-understood-by-all-energy-engineers-there-is-no-right-or-wrong-definition.html ^ Jump up to: a b NIST Chemistry WebBook Jump up ^ Key World Energy Statistics (2005), page 59
- "Carburants et moteurs", J-C Guibet, Publication de l'Institut Français du Pétrole, ISBN 2-7108-0704-1
|




